 In 2012, a collaboration of scientists came together to revamp the electron-scattering method for a more precise measurement of the proton’s charge radius. This discrepancy was dubbed the proton radius puzzle. This result yielded a value that was 4 percent smaller than before, at 0.84184 ±. In 2010, atomic physicists announced results from a new method that measured the transition between energy levels of muons in orbit around lab-made hydrogen atoms that replaced an orbiting electron with a muon, which orbits much closer to the proton and is more sensitive to the proton’s charge radius. When combined, these two methods yielded a radius of 0.8751 ±. In atomic spectroscopy measurements, the transitions between energy levels by electrons are observed (in the form of photons that are given off by the electrons) as they orbit a small nucleus, such as hydrogen (with one proton) or deuterium (with one proton and one neutron). In electron-scattering experiments, electrons are shot at the protons, and the proton’s charge radius is determined by the change in path of the electrons after they bounce off or scatter from the proton.  
Prior to 2010, the most precise measurements of the proton’s charge radius came from two different experimental methods. In addition, the results put an end to speculation that the proton radius puzzle suggested the existence of a new force of nature that acted differently between electrons and muons. This will help scientists better understand these basic components of our universe. This new research gives researchers a more precise way of measuring components of the atom. Nuclear physicists may have now solved the proton radius puzzle with a unique new measurement of the charge radius using a novel electron scattering technique. That’s when a then-new, more sensitive experimental method for measuring the size of the proton by replacing the electron with the heavier muon unexpectedly revealed a value that was 4 percent smaller than obtained from the two previous methods. The “proton radius puzzle” arose in 2010. 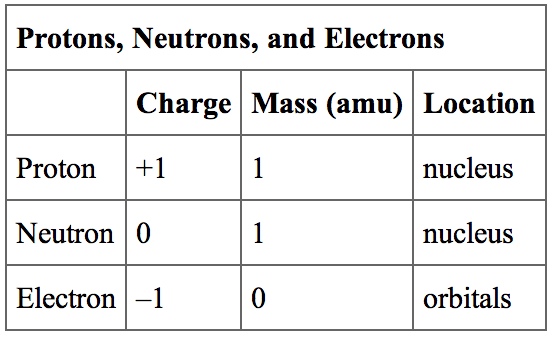
Historically, scientists used two different methods to get an approximate measure of proton size. This makes it hard to measure the size of positively charged protons in nuclei. Textbook pictures make atomic nuclei seem tidy, but they are actually fuzzy clouds of subatomic particles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
